Will UV Light cause Degrade on Detergent
- On Feb, 19, 2021
- admin
- Detergent, Others, Surface Cleaning
People worry about UV light may degrade the detergent but in most cases, the effect may not be significant. The sunlight supports our life and plants growing, but it also comes along with a certain level of UV light. UV is a kind of solar radiation which accessive amount will bring adverse effect to the human body. Such as skin cancer and sunburn effect to our eyes. This is the reason why we are applying sunblock lotion and sunglasses to reduce the UV radiation to our body.
This brings us a new question, will the detergent formulation degrade by the UV light?
Overdose UV light intensity may cause an adverse effect on human skin and eyes, but we are not seeing a degradation in the detergent performance.
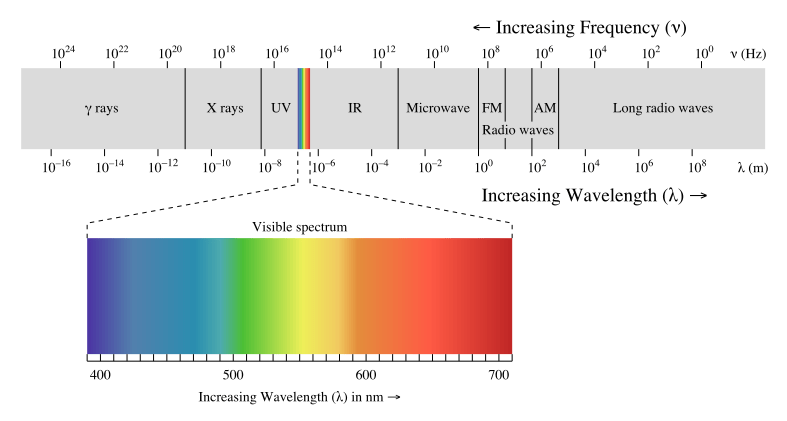
What is UV Light
UV light or ultraviolet light is an energy level comes from 100-400nm wavelength’s electromagnetic radiation. Solar is a great source of UV radiation, apart from there, ultra-violet lamp, arc welding also generate UV light source.
A small dosage of UV light is actually good for human life as it is a good sterilize source. Unfortunately, an overdose of UV light exposure will cause damage to the cell.
So far, we learn that the UV light energy is affecting the organic substance cell and even causing damage or sterilize the substance. This is the reason we are applying sunblock or wearing sunglasses to protect our skin and eyes from excess UV light.
UV Light Effect
Now we come to the nature of a detergent solution, an inorganic compound formulation solution. How should we understand the effect on UV light against the detergent solution? Instead of studying the effect on UV light, we shall broader the view into an overall light source.
In this case, we want to understand the detergent stability after exposing to light. If we are seeing an effect on detergent stability after the photostability test, then we expect to see a degradation of detergent performance as well as the mechanical properties.
UV Light Degrade Exposure Study
A UV light is separated into 3 categories by different wavelength. Namely,
- UV-A (320-400nm wavelength). This is the wavelength of UV light reaching the earth surface. The photostability test study shall focus on this wavelength zone.
- UV-B (280-320nm wavelength). A large portion of UV-B light is absorbed by ozone, only left a small portion of UV-B is reaching the earth surface.
- UV-C (100-280nm wavelength). All of this wavelength of UV light is absorbed by the ozone, and it will not reach the earth surface.
The reader who is interested can always refer to the Ultraviolet Radiation published by WHO.
The Detergent Packing Material
In DST, most of the detergent packed in HDPE drum or pail. Let us compare some differences about these 2 packing materials.
Drum. We are using blue colour 200-L drum for the customer who buys in bulk. This is an HDPE material drum. The blue colourant in the material has minimized light transmission into the drum. However, the blue colourant is not a UV light filter unless a carbon black composite is added.
Furthermore, the HDPE material also can withstand high temperature up to 120C. Even though the HDPE drum may degrade under UV light exposure but it has a long life span under heat. The blue colourant also prevents light transmission into the drum.
Pail. We select the milky translucent HDPE material for 10L or 20L pail. In many practical cases, the user prefers a certain level of “see-through” effect so that they could identify the detergent quickly.
This is also a similar reason why we are seeing a transparent bottle in many household cleaner and detergent.
The Detergent Ingredient
After study the UV light characteristic and packing material, now we move to the detergent ingredient. The industrial used detergent is formulated from a wide selection of surfactant, chelating agent and solvent. Such as Amine Oxide, ethanol and hydrogen peroxide.
The detergent ingredient is an inorganic compound which formulated so that it becomes a stable product. In other words, a reasonable exposure of UV light is less sensitive to the detergent formulation however the heat will do. This also implies that the detergent will be less significant in degradation because expose to UV light.
As we are aware, the photosensitive compound is a material which does not like light. The characteristic may degrade or influence under UV light, for instance, the hydrogen peroxide.
The extensive exposure of UV light will become sufficient energy to generate heat to the detergent. This is also the reason we are asking our user to store the detergent in the shielded area with air ventilation.
Did the UV Light degrade the detergent?
After the above study, we have a better idea about the effect on UV light, the role of packing material and the detergent ingredient. Now, we are coming into the moment to answer whether the UV light may degrade the detergent.
To be frank, we are unable to give you a firm data on the UV effect unless we are performing a comprehensive photostability test on the product. For your information, the photostability test is a testing criterion used in the pharmaceutical industry to determine the drug packaging. The detergent degradation test will give the affirmative result causing by UV light.
However, we are able to make our own judgement base on the above study from various aspect. We can a few conclusion:
- HDPE packaging. Even though the ordinary HDPE is not UV-light filter but it is good to prevent a certain level of light transmitted
- Ingredient. Unless photosensitive compound is used as part of the detergent, most of the inorganic compound is not sensitive to UV light.
- Storage. Instead of UV light, the emphasize shall put in the effect of heat on the detergent. No matter how the detergent shall not exposure to extensive sunlight which eventually will raise the temperature.
Next time while you are getting a detergent, and worry about the degradation effect because of UV light, you could always make your own judgement base on the above criteria. Please write to us if we have unintentionally missed out any part of the information about the UV light effect.






Leave a Comment